Beyond a New Normal
Learning from the COVID-19 Pandemic
In addition to keeping safe, being nimble and resourceful were also required competencies during 2020. Previous investments in infrastructure and system-wide communication enabled a quick transition to remote work for more than 3,000 associates within a matter of weeks — with zero impact, zero systems changes and very limited cost to Nemours Children’s Health. The mission-driven spirit of Nemours associates could not be stifled: from facilitating remote town halls and virtual training to quickly establishing a contactless patient experience in 60+ locations and novel pathways for preventive health measures. Using the mantra: “Never let a crisis go to waste,” key findings, innovative solutions and new alliances are driving our desire to leverage the pandemic experience, and newfound skills, to deliver better health.
Sharing Our Bounty
In March 2020, Nemours Children’s Health Transplant and Immunology Diagnostic Laboratory (TIDL), under the direction of Dr. Paul Fawcett and management of Carrie Paquette-Straub, validated and implemented the gold standard in testing for SARS-CoV2, the first FDA-approved viral test to be offered in a healthcare setting in the state of Delaware. This resource proved so valuable that requests came into the TIDL to make viral testing available to Nemours Children’s Health primary care practices, New Castle County first line responders and St. Francis Hospital. To date, the TIDL has provided COVID-19 testing to more than 15,000 Nemours patients, associates, and first line responders. The TIDL worked closely with Nemours providers to deliver rapid (24-hour) turnaround for screening of patients before surgery, and to test symptomatic and/or exposed Nemours associates, enabling the safe return of patients to Nemours.
In addition to testing, novel approaches to maximize the availability of personal protective equipment became a necessity due to global shortages. Nemours began assembling disposable face shields from foam and film, but they were a temporary solution due to issues with cleaning. A project to create 3D-printed face shields, under the direction of Tariq Rahman, PhD, Director of the Nemours Center for Orthopedic Research & Development, became daunting due to the inability to mass produce quickly. A previous working relationship resulted in a new partnership between Nemours and DuPont Chemical & Delaware Innovation Space to print, cut and assemble face shields at the DuPont Experimental Station. Production quickly scaled up and after two months, this collaboration yielded 3,840 completed, reusable face shields that were assembled and distributed to Delaware hospitals and some nursing homes.
Nemours Children’s Health continued to share its resources with the community. By years end, Nemours’ temperature-controlled storage capabilities in our laboratories across Florida were used to store COVID-19 vaccines for community distribution in Central and Northeast Florida.
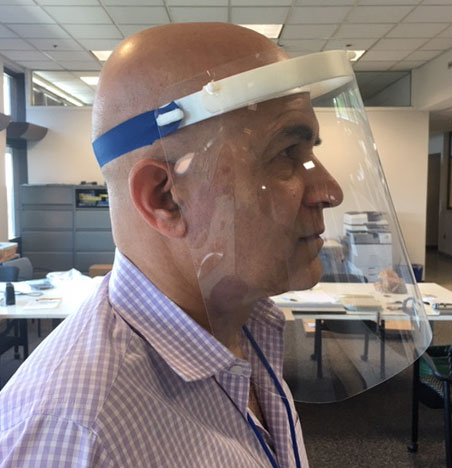
Tariq Rahman, PhD, Director of the Nemours Center for Pediatric Orthopedic Research & Development, models the 3D-printed, reusable face shields assembled in partnership with DuPont Chemical during spring 2020.

Nemours TIDL validated the first FDA-approved viral test for SARS-CoV2 to be offered in a healthcare setting in Delaware and provided it to more than 15,000 patients, associates, and first-line responders. Read more »
1,071% increase in telehealth visits in 2020;
maintained 4.9/5-star provider rating
Disseminating Advice, Comfort & Knowledge
Pediatric experts across Nemours Children’s Health, including specialty doctors and clinicians from nursing and behavioral health, shared valuable information freely throughout the year via webinars, online chats and other virtual forums. Their aim was to help parents, kids, educators and others stay safe — physically and emotionally. With a strong desire to connect individuals to their whole health, stay aware of implications specific to physical disease, and cope with the ever-present range of emotions and feelings of isolation, more than a dozen free webinars and virtual events were attended by thousands from our communities.
These online events covered topics such as “Living with Congenital Heart Disease in the Age of COVID-19,” “School Health Trainings: Child Sexual Abuse & Treatment,” “COVID-19 and the Holidays: Keeping Your Family Safe & Healthy,” and sometimes a little fun like the “Williams Syndrome Family Dance Party & Talent Show.”
Early in the pandemic, Nemours KidsHealth and the Nemours Office of Health Equity & Inclusion quickly created, translated and ensured community reflection for COVID-19 resources hosted on KidsHealth.org, the world’s most visited website for trusted children’s health information. The materials and video guides, which began publishing in January 2020, were directed at families around the world and included topics like “Know Your Symptoms,” “Stop the Spread,” “Calm Your Family,” “How to Protect Babies & Toddlers” and “MIS-C in Children.” With more than 40 million views, the COVID-related content serves as a critical form of education for a vast number of families.

Getting Ready, Staying Safe
Just as Nemours Children’s Health had to plan for its own safe, phased return to in-person activities, school leaders, parents and community health organizations were looking for evidence-based information to determine their safest courses of action during the summer and fall months of 2020. Nemours Children’s Health Infectious Disease specialists Dr. Kenneth Alexander, MD, PhD, and Adriana Cadilla, MD, jointly and individually collaborated with Central Florida’s superintendents of schools to offer medical advice and presentations on ways the schools could reopen safely during the pandemic. Leveraging Nemours experts access to science-based information and the latest findings, as the novel coronavirus evolves, they continue to serve on task forces for school districts in Orange and Osceola counties to help monitor community infection rates in schools. Their partnership resulted in being named Osceola County School Board’s “District Partners of the Year” in addition to recognition as Heart of Florida United Way, “Change Maker’s of the Year.”
Drs. Alexander and Cadilla also partnered with infectious disease colleagues outside of Nemours to author “Considerations for Teachers and Staff,” that drew on the latest science and experiences with COVID-19 in children’s hospitals to inform the recommendations for schools. Their hope was to guide school districts’ leadership in their decision-making on how to minimize the risk of exposure in the school environment.
Additionally, Nemours infectious disease professionals collaborated with their peers to devise a tool for providers to confidently and uniformly discuss with parents the advantages and disadvantages of in-person and virtual education.
The organizations recognized serve as a model for how the community and a school system can form strategic partnerships that work in support of students and schools as we become stronger together. — Debra Pace, Superintendent, Osceola County School District
Preparing for a New Day: Continuing to Learn from COVID-19
Nemours Cardiac Center to Participate in New MIS-C Study
The Nemours Cardiac Center, under the direction of Shubhika Srivastava, MD, Chief of Cardiology, Nemours Children’s Hospital Delaware, will be one of the research sites participating in a new study managed by HealthCore to help understand and combat the pediatric public health emergency for MIS-C, related to COVID-19. The study has been named the MUSIC study, short for the Long-TerM OUtcomes after the Multisystem Inflammatory Syndrome In Children. It will be the first study to provide long-term outcome data on the newly defined multisystem inflammatory syndrome in children, or MIS-C, which usually occurs in otherwise healthy children and has disproportionately affected Black and Hispanic/Latino children.
Data Driven: The National COVID Cohort Collaborative
The National COVID Cohort Collaborative (N3C) is a partnership among the Clinical and Translational Science Awards Program hubs, the National Center for Data to Health, the Clinical and Translational Research networks (CTRs), distributed clinical data networks (PCORnet, OHDSI, ACT/i2b2, TriNetX), and other partner organizations, with overall stewardship by NIH’s National Center for Advancing Translational Sciences. The aims are to improve the efficiency and accessibility of COVID-19 research by providing (1) a centralized, harmonized and aggregated patient database and (2) an analytical ecosystem to collaborate and share insights.
The data set includes demographics, symptoms, lab test results, procedures, medications, medical conditions, physical measurements, and more. Eight CTRs (West Virginia, Northern New England, Rhode Island, Mississippi, Oklahoma, Nebraska, Louisiana, and Delaware) are contributing data sets to this national collaboration, which includes data from rural and urban centers of IDeA states. These data sets should provide key information for CTR investigators as well as inform national researchers and policymakers.

Nemours is contributing data sets to the National COVID Cohort Collaborative to inform national researchers and policymakers. Read more »
Additional research grants have been awarded to Nemours investigators to further understanding of the physical, emotional and social impacts of the pandemic, including:
- Dr. Maricar Diaz: “A Feasibility Study to Measure Protection Offered by a COVID-19 Intubation Enclosure”
- Dr. Anne Kazak: “How does the pandemic affect families? The COVID-19 Exposure and Family Impact Scale (CEFIS)”
- Dr. Deepika Thacker: “Early prevalence of SARS-CoV-2 antibody in children presenting with Kawaski diease/toxic shock syndrome”
- Dr. Carissa Baker-Smith: “Early Experience of Telehealth in Pediatric Cardiology During the COVID-19 Pandemic”
- Dr. Ly Phan: “The Impact of COVID-19 on Families and Pediatric Healthcare Delivery”
- Dr. Erin Crowgey: “Enhancing COVID-19 diagnostics and genomic surveillance using innovative molecular biology techniques”